|
Interesting Facts about Phosphorus. When Hennig Brand discovered phosphorus, he became the first person in history to discover an element. The Saturated Hydrocarbons, or Alkanes. Compounds that contain only carbon and hydrogen are known as hydrocarbons. Those that contain as many hydrogen atoms as. Appearance and Characteristics. Harmful effects: Silicon is not known to be toxic, but if breathed in as a fine silica/silicate dust it may cause chronic respiratory. More Practice With Naming. Before naming a compound, you have to figure out what kind of compound it is. We will consider three types.   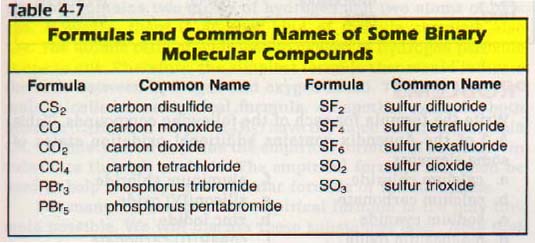
As the name of a non- metallic chemical element, it is recorded from 1. It was discovered in 1. Henning Brand, merchant and alchemist of Hamburg, who derived it from urine. Lavoisier demonstrated it was an element in 1. 
Chemistry And The Aquarium: Phosphorus: Algae's Best Friend — Advanced Aquarist. Phosphorus is one of the basic building blocks of living matter. At p. H 8. 1, seawater contains 0. H2. PO4- , 7. 9 % HPO4. PO4. 3- . At higher p. H the equilibrium shifts toward more PO4. HPO4. 2- . The shift in distribution with p. H may seem esoteric, but it actually has important implications for such things as the binding of phosphate to rock and sand. There are a number of reasons for this difference between salt water and fresh water that involve the effects of other ions in the seawater on the phosphate (such as calcium and magnesium ion- pairs), and these have been described previously. Other Forms of Inorganic Phosphate. Phosphorus can also take other inorganic forms, such as the polyphosphates which are rings and chains of phosphate ions strung together by P- O- P bonds. Consequently, while these products may do a fine job of reducing inorganic orthophosphate, they may not help an algae problem that is caused primarily by organic phosphates. 
A final point is that organic phosphates will not be detected by most test kits. Since phosphorus is required for growing tissue, it is mandatory that there be some phosphorus source for corals growing in a reef tank. The water in many reef tanks will be supersaturated with respect to this material, as the equilibrium saturation concentration in normal seawater is only 0. As with Ca. CO3, the precipitation of Ca. PO4)2in seawater may be limited by kinetic factors more than equilibrium factors, so it is impossible to say how much might precipitate under reef tank conditions (without, of course, somehow determining it experimentally). This precipitation may be especially likely where calcium and high p. 
H additives (like limewater) enter the tank water. The locally high p. H converts much of the HPO4. PO4. 3- . Combined with the locally high calcium, the locally high PO4. Ca. 3(PO4)2 to unstable levels, causing precipitation. Likewise, phosphate can precipitate onto the surface of calcium carbonate, such as onto live rock and sand. All of the other species tested gave similar results (plus or minus a factor of 2). 
We have already encountered some chemical formulas for simple ionic compounds. A chemical formula is a concise list of the elements in a compound and the ratios of. Phosphorus is found in most food because it is a critical constituent of all living organisms. Dairy foods, cereal products, meat, and fish are. Naming Compounds. Naming Compounds. More Practice With Naming. Before naming a compound, you have to. 
We will consider three types: Ionic Compounds Without a Transition Metal. Ionic compounds are formed when a metal gives up its electrons to a non- metal. But there are. different sets of rules for transition metals. A transition metal is an element with an atomic number of 2. In such a case the total charge of the (+) and. ZERO. Example: What is the correct formula for calcium phosphide? Here, we have to. Calcium, if you recall, typically loses two electrons to get a. To get a sum of. zero, we need three Ca+2 ions and two P- 3 for a total of. So the answer is. K+1 and O- 2 give K2. O aluminum bromide Al+3. 
Br- 1 yield Al. Br. To get a sum charge of ZERO, we need two. Al. 2(SO4)3. Ionic Compounds With a Transition Metal. The only. difference here is that we have to specify the charge of the transition metal. Roman numeral, and keep in mind that a transition metal is an. Roman numeral. Charge. I+1. II+2. III+3. IV+4. V+5. VI+6. The reason we do. Roman numerals nor to imitate. Because transition metals can assume more than one charge, we. Example: manganese(II) oxide contains Mn+2 and. O- 2. So we just need one of each and the formula becomes Mn. O. To go backwards, we need to figure out the charge of the transition. Example: What is the correct name. Cr. Cl. 3 ? The charge of Cr is unknown. The sum of the charges has top be zero, so: x. These are formed from non- metals. Because there are many sharing possibilities between two. For this we use. a set of prefixes: Prefix. Number of atomsmono. The only time we. Examples: CO = carbon monoxide ( note. CO2 = carbon. dioxide dinitrogen. N2. O5. Metals do not form covalent compounds. Summary: 1. Write formulae for the following: a. Understanding phosphorus fertilizers : Nitrogen : University of Minnesota Extension. Extension > Agriculture > Nutrient Management > Phosphorus > Understanding phosphorus fertilizers. By George Rehm, Michael Schmitt, John Lamb, Gyes Randall, and Lowell Busman Copyright . Several phosphate fertilizers can be used to meet the phosphorus (P) requirements of Minnesota's crops. This extension folder is designed to: give a general description of the processes used in the manufacture of commercial phosphate fertilizers; describe differences and similarities for the various materials. Manufacturing commercial phosphate fertilizer. Rock phosphate is the raw material used in the manufacture of most commercial phosphate fertilizers on the market. In the past, ground rock phosphate itself has been used as a source of P for acid soils. The process used in the manufacture of various phosphate fertilizers. The manufacture of most commercial phosphate fertilizers begins with the production of phosphoric acid. A generalized diagram showing the various steps used in the manufacture of various phosphate fertilizers is provided in Figure 1. Phosphoric acid is produced by either a dry or wet process. In the dry process, rock phosphate is treated in an electric furnace. This treatment produces a very pure and more expensive phosphoric acid (frequently called white or furnace acid) used primarily in the food and chemical industry. Fertilizers that use white phosphoric acid as the P source are generally more expensive because of the costly treatment process. The impurities which give the acid its color have not been a problem in the production of dry fertilizers. The phosphate concentration in superphosphoric acid usually varies from 7. The P in this acid is present as both orthophosphate and polyphosphate. Polyphosphates consist of a series of orthophosphates that have been chemically joined together. Upon contact with soils, polyphosphates revert back to orthophosphates. The liquid, 1. 0- 3. The 1. 0- 3. 4- 0 can be mixed with finely ground potash (0- 0- 6. The P in these products is present in both the orthophosphate and polyphosphate form. The P present in these two fertilizers is present in the orthophosphate form. More importantly, the processes used have no effect on the availability of P to plants. Phosphate Fertilizer Terminology Because of the number of products on the market, the selection of a phosphate fertilizer can be confusing. An explanation of some terminology may help to avoid some of the confusion. Some important terms are: Water- Soluble– Fertilizer samples analyzed by a control laboratory are first placed in water and the percentage of the total phosphate that dissolves is measured. This percentage is referred to as water- soluble phosphate. Citrate- Soluble –The fertilizer material that is not dissolved in water is then placed in an ammonium citrate solution. The amount of P dissolved in this solution is measured and expressed as a percentage of the total in the fertilizer material. Phosphate measured with this analytical procedure is referred to as citrate- soluble. Available –The sum of the water- soluble and citrate- soluble phosphates is considered to be the percentage that is available to plants and is the amount guaranteed on the fertilizer label. Usually, the citrate- soluble component is less than the water- soluble component. Table 1. Percentages of water- soluble and available phosphate in several common fertilizer sources. P2. O5. P2. O5 Source. N (%)Total (%)Available (%)Water soluble* (%)Superphosphate (OSP)0. Concentrated Superphosphate (CSP)0. Monoammonium Phosphate (MAP)1. Diammonium Phosphate (DAP)1. Ammonium Polyphosphate (APP)1. Rock Phosphate. 03. Water- soluble data are a percent of the total P2. O5 Source: Ohio Agronomy Guide. Organic Phosphorus Sources Organic P fertilizers have been used for centuries as the P source for crops. Even with the advent of P fertilizer technology processes, organic P sources from animal manures—including composts—and sewage sludge are still very important. From a fertilizer/nutrient management perspective, the major differentiating factor is the availability of P. Then an availability coefficient should be used to determine the available P as a portion of the reported total P. Therefore, if a producer has a P recommendation for 3. A of P2. O5 , applying approximately 6. DAP) or 6 tons of 1. P coefficient) should provide equivalent results. Essentially, all of the inorganic P is in the orthophosphate form, which is the form taken up by growing plants. Diet fed to the animal has some control on this chemical make- up. Consider P feed supplements and the fact that many of these could be considered P fertilizers as well. Generally, 4. 5- 7. P is inorganic P. Much of the organic P is easily decomposable in the soil, but factors such as temperature, soil moisture, and soil p. H all have a bearing on the P mineralization rate. Most animal manure research interpretations indicate that approximately 6. P is available to crops in the first year. Due to the chemical composition of other organic P sources such as bone meal, lesser amounts of plant available P compared to total P are expected. Phosphorus Source Issues Some of the most frequently asked questions about phosphate fertilizer are discussed in the paragraphs that follow. Plant nutrient use in both liquid and dry fertilizers is affected by such factors as method of application, crop and root growth characteristics, soil test levels, and climatic conditions. The amount of water in a fluid fertilizer is insignificant compared to the water already present in the soils. Therefore, P in liquid P sources is not more available than P in dry materials — even in a dry year. The selection of a liquid or dry P source should be based on adaptation to the farmer's operation and economics. The phosphorus in the phosphoric acid used to make most dry phosphate fertilizers as well as a few liquids is in the orthophosphate form. This process does not convert 1. This conversion is rapid and, with normal soil temperatures, can be complete in days or less. This conversion process is enhanced by an enzyme called pyrophosphatase, which is abundant in most soils. Because water is removed in the manufacturing process, these materials have a higher analysis than materials in which the phosphate is in the orthophosphate form. The polyphosphate liquids are also more convenient for the fertilizer dealer to handle and allow for the formulation of blends that are not possible with the orthophosphate liquids. The results shown in Table 2 are typical of the results obtained from several trials. The influence of P source on corn yield. P2. O5 Applied. P Source. Polyphosphate. Orthophosphatelb./acrebu./acre. Source: Nebraska Soil Test P: Low The yields shown in Table 2 are averages from five sites where the soil p. H was in excess of 7. Similar results from other studies have been reported throughout the Corn Belt. From an academic perspective, monoammonium phosphates (MAP) create a more acidic zone around each fertilizer granule, whereas diammonium phosphates (DAP) create a basic zone. Thus, in high p. H soils, it can be theorized that using MAP- based fertilizers should be better than DAP because the acid- producing fertilizer would offset the calcareous soils. An additional concern regarding MAP or DAP selection, aside from soil p. H, is potential ammonia toxicity to germinating seeds in dry soils. In applying the recommended amount of P in a drill- row or pop- up fertilizer placement, DAP will contain approximately 6. N, which may be a potential injury risk. However, since agronomic studies and economic data indicate no crop yield differences, it can be concluded that fertilizer selection should be made on traditional factors such as nutrient content, price, availability, etc. Summary There are many P- containing fertilizers to satisfy any P recommendation. Inorganic P fertilizers have evolved over the last several decades into a refined, predictable product. Certain situations have provided certain instances where one product or the other was superior. However, phosphorus fertilizer recommendations are the same regardless of the phosphate fertilizer source.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
November 2017
Categories |
 RSS Feed
RSS Feed
